16.1 Thermal Energy and Matter
Questions About Thermal Energy & Matter1. Which has more thermal energy, a cup of tea or a pitcher of juice?
2. Which has a higher temperature, a cup of tea or a pitcher of lemonade. 3. Which has more thermal energy, a cup of hot tea or a cup of cold tea? |
|
Work and Heat
*Heat is the transfer of thermal energy from one object to another as the result of a difference in temperature.
*Heat flows spontaneously from hot objects to cold objects.
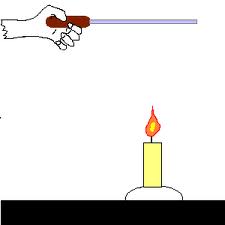
*Friction produces heat.
*Heat flows spontaneously from hot objects to cold objects.
*Friction produces heat.
Temperature
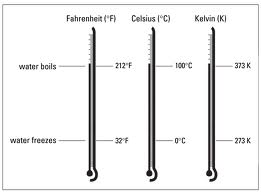
* What is temperature? Temperature is a measure of how hot or cold an object is compared to a reference point.
* On the Celsius scale, the reference points for temperature are teh freezing and boiling points of water.
*When an object heats up, it's particles move faster, on average, mass increases, and temperature increases.
* On the Celsius scale, the reference points for temperature are teh freezing and boiling points of water.
*When an object heats up, it's particles move faster, on average, mass increases, and temperature increases.
Boiling points on Celcius scale
Thermal Energy
*What is thermal energy?
The toal potential and kinetic energy of all the particles in an object.
*Thermal energy depends upon the mass, temperature, and phase of matter.
The toal potential and kinetic energy of all the particles in an object.
*Thermal energy depends upon the mass, temperature, and phase of matter.
Thermal Contraction
Thermal Expansion
Specific Heat
*The amount of heat needed to raise the temperature of one gram of material by one degree Celsius is called specific heat.
*Why are you most likely to burn yourself on a metal toy than on a plastic toy if both have been sitting in the sun?
The iron in the metal has a lower specific heat than the plastic toy. If equal masses of iron and plastic absorb the same heat, the iron's temperature rises more.This shows that two items can have the same temperature but different thermal energies.
*Why are you most likely to burn yourself on a metal toy than on a plastic toy if both have been sitting in the sun?
The iron in the metal has a lower specific heat than the plastic toy. If equal masses of iron and plastic absorb the same heat, the iron's temperature rises more.This shows that two items can have the same temperature but different thermal energies.
Measuring Heat Changes
*What device is uded to measure changes in thermal energy?
Calorimeter.
*A calorimeter uses the principle that heat flows from a hotter object to a colder object until both reach the same temperature.
Calorimeter.
*A calorimeter uses the principle that heat flows from a hotter object to a colder object until both reach the same temperature.
16.2 Heat and Thermodynamics
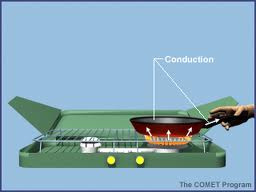
Conduction
Transfer of Thermal Energy
Conduction: transfar of thermal energy with no net transfer of matter.
Ex: Frying pan handle heats up.
1. The transfer of thermal energy with no overall transfer of matter is called conduction.
2. Why is conduction slower in gases than in liquied and solids?
Becasue the particles in a gas collide less often.
3, Conduction is faster in metals than in other solids because metals have free electrons that transfer thermal energy.
Ex: Frying pan handle heats up.
1. The transfer of thermal energy with no overall transfer of matter is called conduction.
2. Why is conduction slower in gases than in liquied and solids?
Becasue the particles in a gas collide less often.
3, Conduction is faster in metals than in other solids because metals have free electrons that transfer thermal energy.
Type of MaterialThermal Conductors
Thermal Insulator
| Quality of ConductionConducts thermal energy well
Conducts thermal energy poorly
| Two ExamplesCopper; Aluminum
Wood, Plastic
|
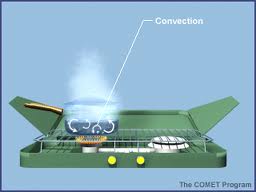
Convection
*The transfer of energy when particles of a fluid move from one place to another is called convection.
*Why is temperature higher at teh bottom of an over?
When air at teh bottom of the oven heats up, it expands and becomes less dense than the surrounding air. Due to the difference in density, the hot air rises. The rising air cools as it moves away from the heat source,. As a result, the coolest air is at the top of the oven.
*When a fluid curculates in a loop as it alternately heats up and cools down, a convection current occurs.
* Examples of convection curruents:
-ocean currents
-weather systems
-movements of hot rock in Earth's interior.
*Why is temperature higher at teh bottom of an over?
When air at teh bottom of the oven heats up, it expands and becomes less dense than the surrounding air. Due to the difference in density, the hot air rises. The rising air cools as it moves away from the heat source,. As a result, the coolest air is at the top of the oven.
*When a fluid curculates in a loop as it alternately heats up and cools down, a convection current occurs.
* Examples of convection curruents:
-ocean currents
-weather systems
-movements of hot rock in Earth's interior.
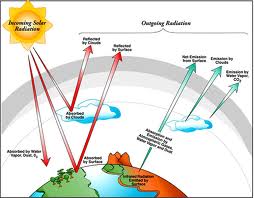
Radiation
*The transfer of energy by waves moving through space is called radiation.
*Energy is transferred by waves.
*The farther away you are from a radiating object, the less radiation you recieve.
*All objects radiate energy.
*Energy is transferred by waves.
*The farther away you are from a radiating object, the less radiation you recieve.
*All objects radiate energy.
Thermodynamics
*Thermodynamics is the study of conversions between and other forms of energy.
*The first law of thermodynamics states that energy is conserved.
*The second law of thermodynamics states that thermal energy can flow from colder objects to hotter objects only if work is done on the system.
*The first law of thermodynamics states that energy is conserved.
*The second law of thermodynamics states that thermal energy can flow from colder objects to hotter objects only if work is done on the system.